Mechanochemistry is a widespread synthesis technique in all areas of chemistry. Various materials have been synthesized by this technique when the classical wet chemistry route is not satisfactory. Characterization of the reaction mixture is however much less accessible than in solutions.
Recently, in situ observations of mechanochemical reactions have been achieved by X-ray diffraction and Raman spectroscopy. Solid-state reactions can be directly tracked, revealing phase transitions and other material transformations during synthesis in a ball mill jar. This technique has become increasingly popular in different fields of mechanochemistry.
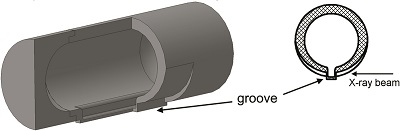
As the X-rays go through the entire jar, the diffraction patterns present a high background due to the scattering from the thick walls of the jar. Also, broad diffraction peaks are expected from the sample as a result of probing a large sample area covering the entire jar. An extra complexity arises from diffraction on the milling balls.
Tumanov et al. [(2017). J. Appl. Cryst. 50. doi:10.1107/S1600576717006744] reasoned that these issues can be resolved by modifying the geometry and material of the milling jar. But, making a jar with a complex geometry using traditional production techniques is complicated, especially at the stage of creating a prototype, when introducing changes into a design should be facile. For this reason they decided to use a 3D printer for the purpose. They show how this useful production tool can quickly make milling jars optimized for improved background, absorption and angular resolution in X-ray powder diffraction experiments; the jars are also more resistant to solvents compared with standard acrylic jars. 3D printing allows for low-cost fast production on demand.
Source files for printing the jars are available as supporting information for the paper.
